OUR
PRODUCTS
Global No. 1 in the field of in vitro diagnosis
-
POINT-OF-CARE
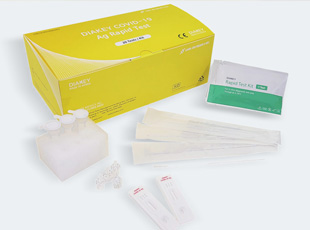
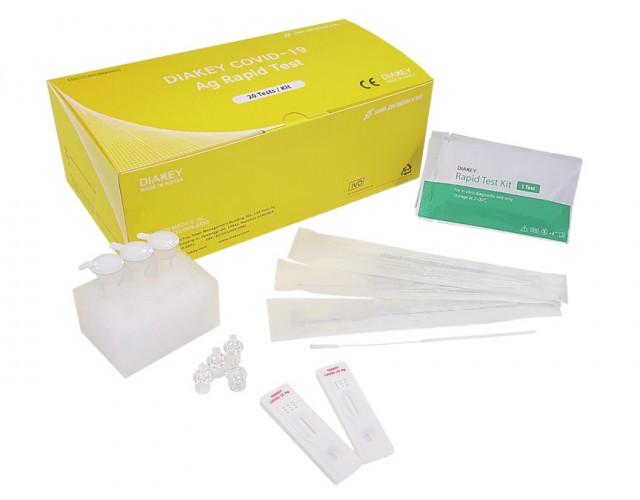
DIAKEY COVID-19 Ag Rapid Test
DIAKEY COVID-19 Ag Rapid Test
-
POINT-OF-CARE
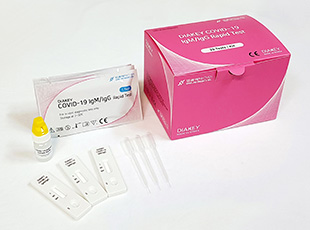
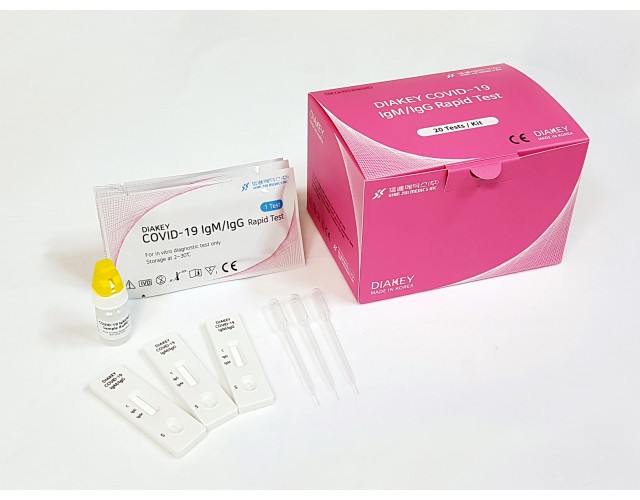
DIAKEY COVID-19 IgM/IgG Rapid Test
DIAKEY COVID-19 IgM/IgG Rapid Test
-
ELISA Instrument
ELISA Instrument
EIA Microplate Washer

-
ELISA Instrument
ELISA Instrument
EIA Spectrophotometer

-
ELISA Instrument
ELISA Instrument
EIA Microplate Shaking Incubator

-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY NSE ELISA
-
ENZYME IMMUNOASSAY
HORMONES & OTHERS
DIAKEY FSH ELISA
-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY Ferritin ELISA
-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY Free T3 ELISA
-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY Free T4 ELISA
-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY TSH ELISA

-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY T4 ELISA
-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY PSA ELISA

-
RADIO IMMUNOASSAY
TUMOR MARKERS
DIAKEY CA19-9 ELISA

-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY Anti-HAV IgG ELISA
-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY T3 ELISA
-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY CA125 ELISA
-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY CEA ELISA

-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY AFP ELISA

-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY H.Pylori IgG ELISA
-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY Anti-HIV 1/2 ELISA
-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY Anti-HCV 3.0 ELISA
-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY H.Pylori IgG ELISA
-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY Anti-HBs ELISA
-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY HBsAg ELISA
-
POINT-OF-CARE
DIAKEY DOA6 RAPID
DIAKEY DOA6 RAPID
-
POINT-OF-CARE
DIAKEY Syphilis RAPID
DIAKEY Syphilis RAPID
-
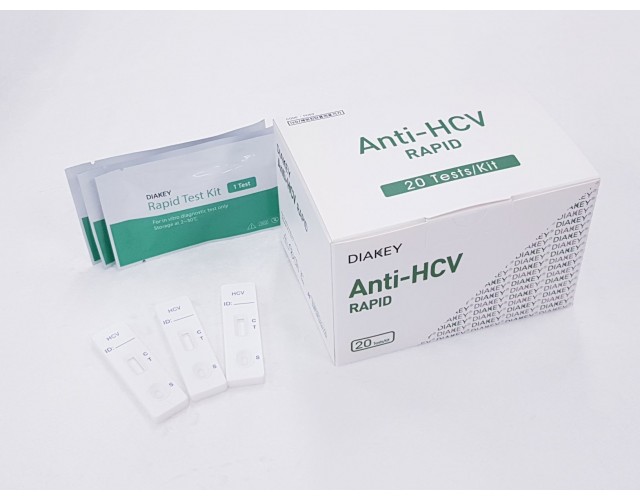
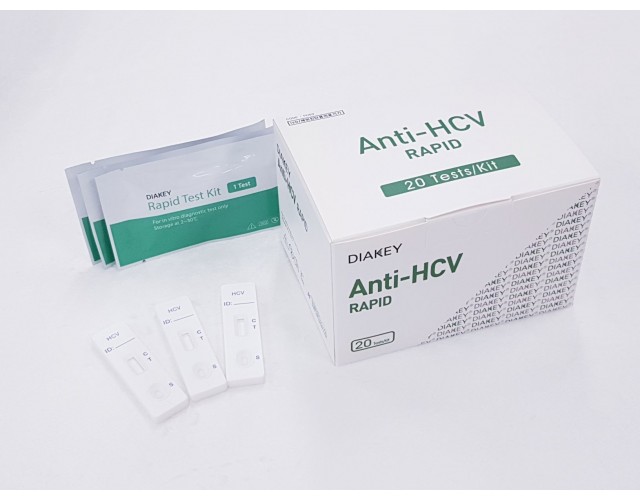
POINT-OF-CARE
DIAKEY Anti-HCV RAPID
DIAKEY Anti-HCV RAPID
-
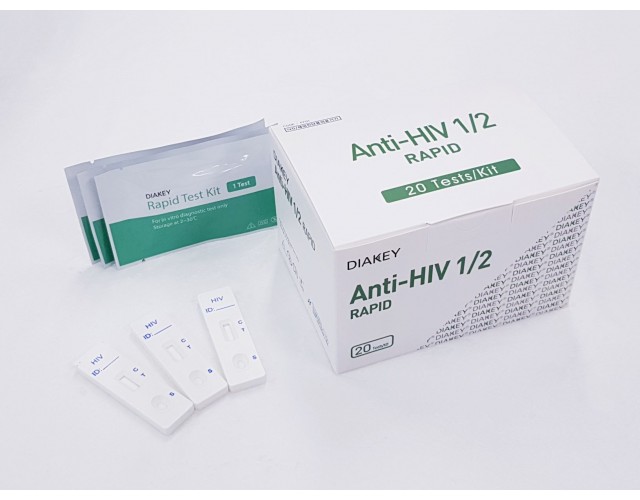
POINT-OF-CARE
DIAKEY Anti-HIV 1/2 RAPID
DIAKEY Anti-HIV 1/2 RAPID
-
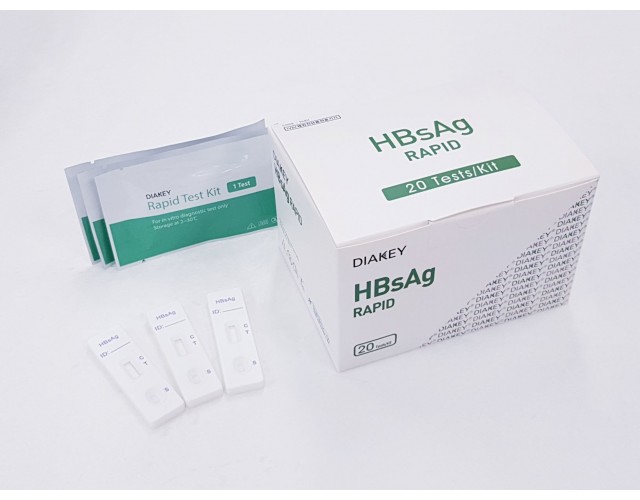
POINT-OF-CARE
DIAKEY HBsAg RAPID
DIAKEY HBsAg RAPID
-
RIA Instrument
RIA Instrument
MESSIAH DS-8150
-
polymerase chain reaction (PCR)
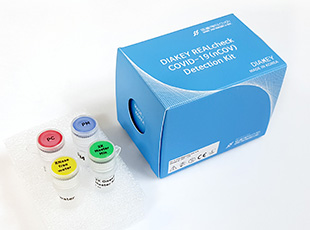
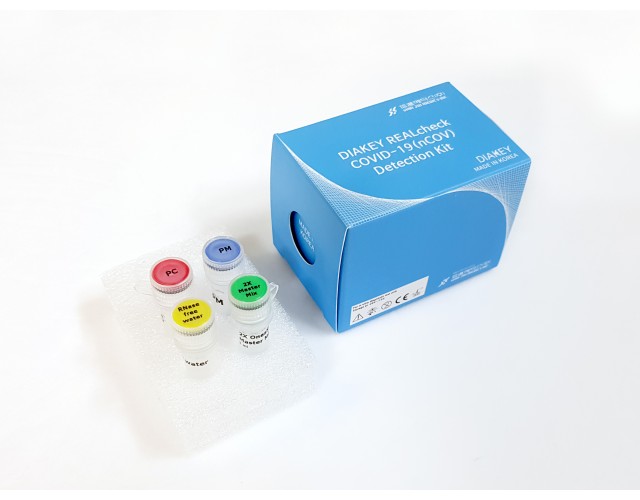
DIAKEY REALcheck COVID-19(nCOV) Detection Kit
DIAKEY REALcheck COVID-19(nCOV) Detection Kit
-
polymerase chain reaction (PCR)
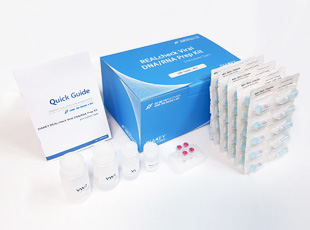
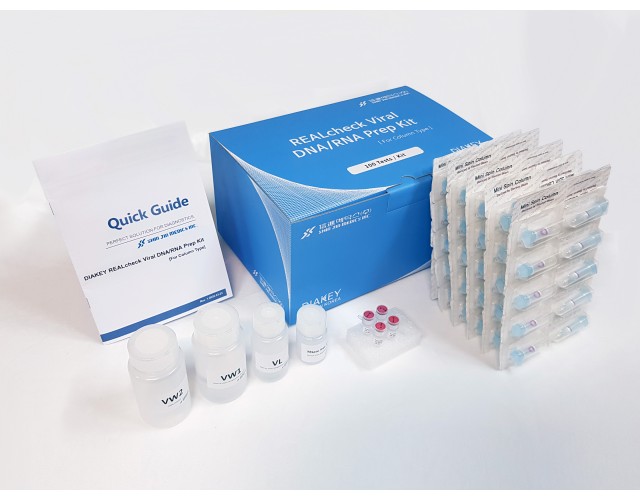
DIAKEY REALcheck Viral DNA/RNA Prep Kit
DIAKEY REALcheck Viral DNA/RNA Prep Kit
-
RADIO IMMUNOASSAY
HORMONES & OTHERS
C-peptide IRMA Tube II
-
RADIO IMMUNOASSAY
HORMONES & OTHERS
Insulin IRMA Tube II
-
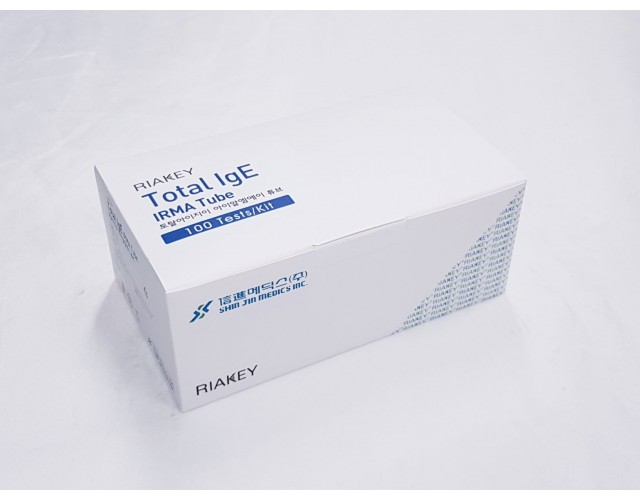
RADIO IMMUNOASSAY
HORMONES & OTHERS
Total IgE IRMA Tube
-
RADIO IMMUNOASSAY
HORMONES & OTHERS
β-hCG IRMA Tube

-
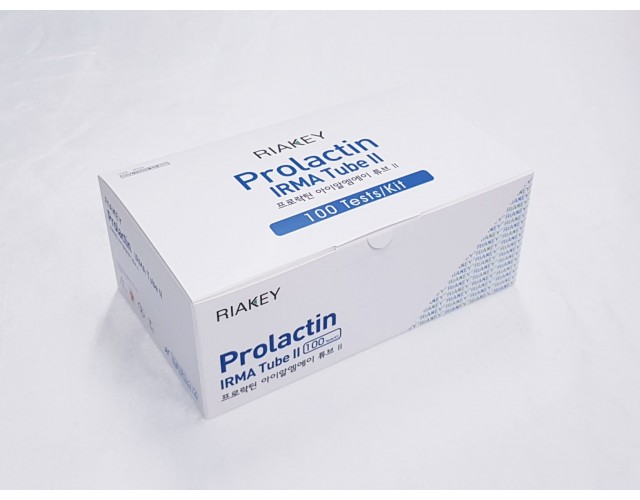
RADIO IMMUNOASSAY
HORMONES & OTHERS
Prolactin IRMA Tube II
-
RADIO IMMUNOASSAY
HORMONES & OTHERS
FSH

-
RADIO IMMUNOASSAY
TUMOR MARKERS
SCC

-
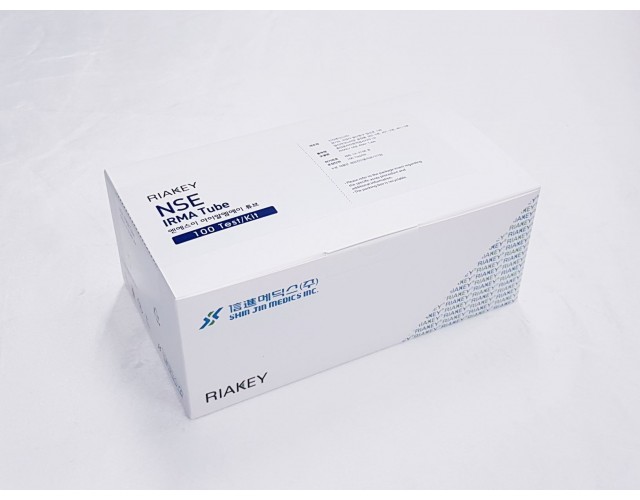
RADIO IMMUNOASSAY
TUMOR MARKERS
NSE
-
RADIO IMMUNOASSAY
TUMOR MARKERS
FERRITIN

-
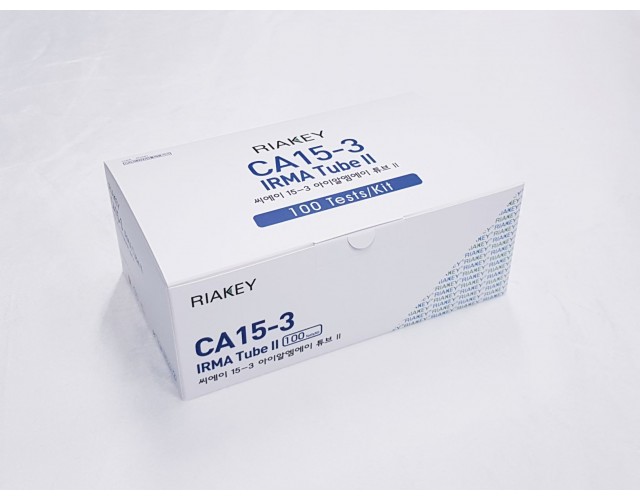
RADIO IMMUNOASSAY
TUMOR MARKERS
CA 15-3
-
RADIO IMMUNOASSAY
TUMOR MARKERS
CA 19-9(2step)
-
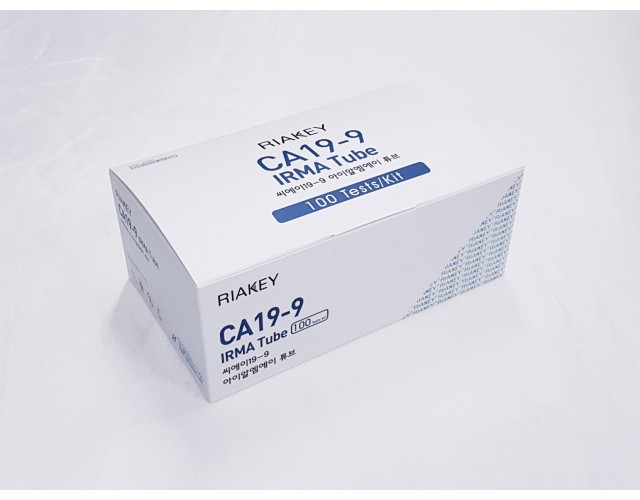
RADIO IMMUNOASSAY
TUMOR MARKERS
CA 19-9(1step)
-
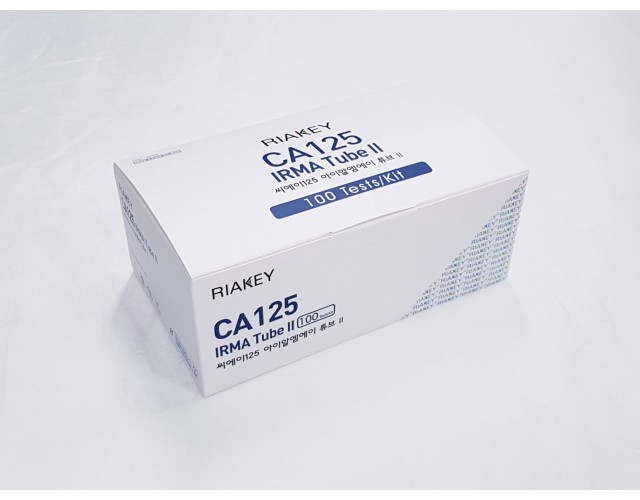
RADIO IMMUNOASSAY
TUMOR MARKERS
CA125 IRMA Tube II
-
RADIO IMMUNOASSAY
TUMOR MARKERS
Ultra PSA

-
RADIO IMMUNOASSAY
TUMOR MARKERS
Free PSA

-
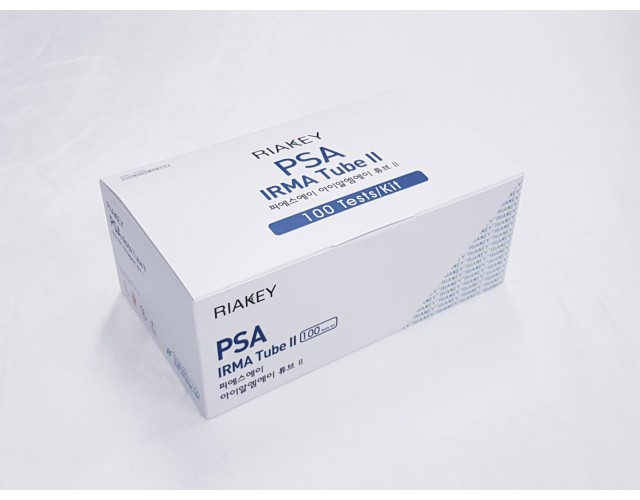
RADIO IMMUNOASSAY
TUMOR MARKERS
PSA
-
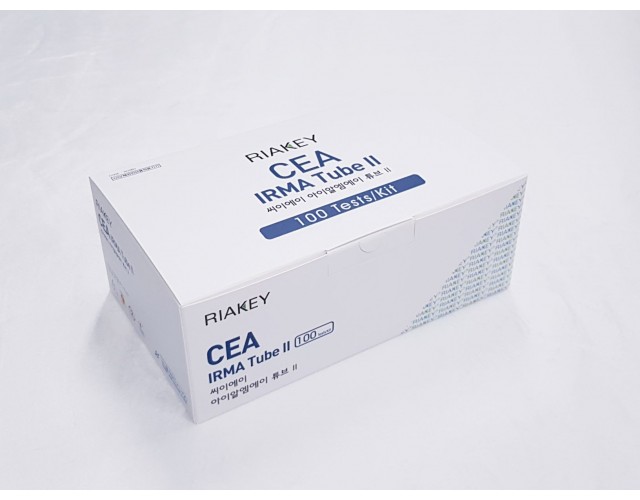
RADIO IMMUNOASSAY
TUMOR MARKERS
CEA
-
RADIO IMMUNOASSAY
TUMOR MARKERS
AFP
-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
Anti-Tg
-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
Tg-s
-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
RIAKEY TSH IRMA Tube II
-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
T3-Uptake
-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
Free T4
-
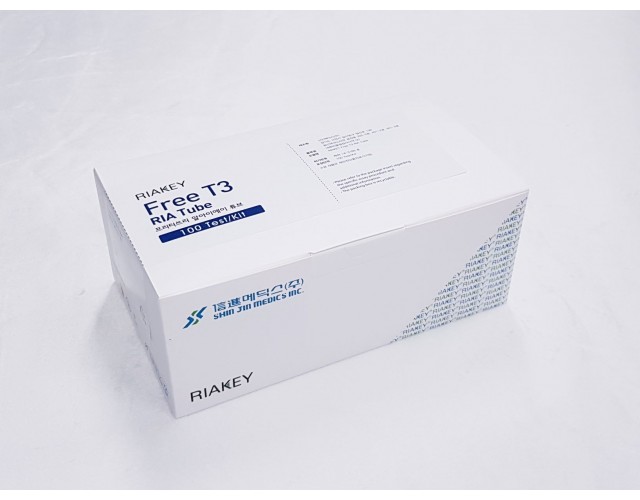
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
Free T3
-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
T4

-
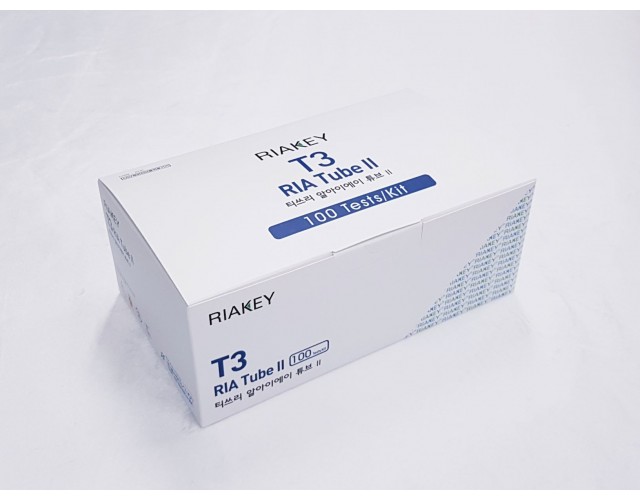
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
T3
-
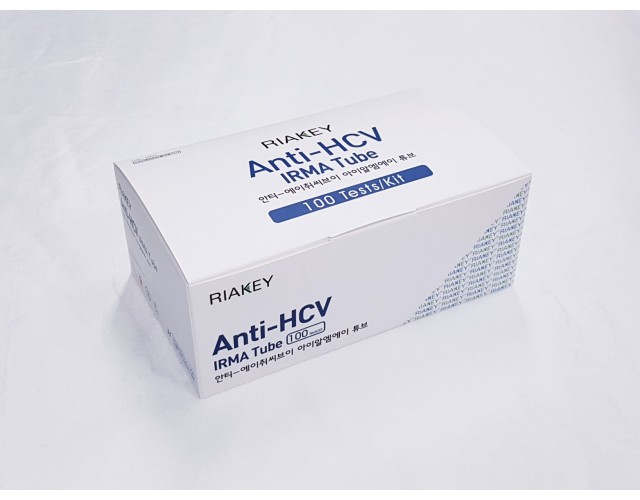
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HCV
-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HAV lgM
-
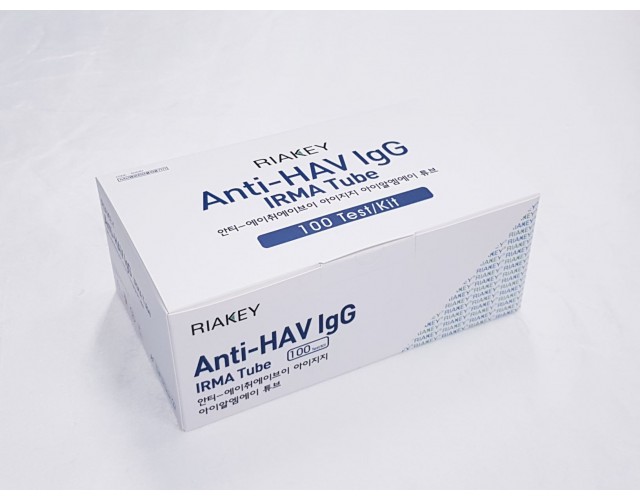
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HAV lgG
-
RADIO IMMUNOASSAY
HEAPATITIS
HBeAg/Ab
-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HBc (RIA)
-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HBc lgM
-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HBc lgG
-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HBs
-
RADIO IMMUNOASSAY
HEAPATITIS
HBsAg
-
RIA Instrument
RIA Instrument
MESSIAH SI-600 MK2

-
RADIO IMMUNOASSAY
HEAPATITIS
RIAKEY AFP IRMA Tube II
-
RIA Instrument
RIA Instrument
MESSIAH TW-300 MK2

-
RIA Instrument
RIA Instrument
MESSIAH Gamma Counter

-
RIA Instrument
RIA Instrument
MESSIAH R-4200
-
RADIO IMMUNOASSAY
TUMOR MARKERS
DIAKEY CA19-9 ELISA

-
RADIO IMMUNOASSAY
HORMONES & OTHERS
C-peptide IRMA Tube II

-
RADIO IMMUNOASSAY
HORMONES & OTHERS
Insulin IRMA Tube II

-
RADIO IMMUNOASSAY
HORMONES & OTHERS
Total IgE IRMA Tube

-
RADIO IMMUNOASSAY
HORMONES & OTHERS
β-hCG IRMA Tube

-
RADIO IMMUNOASSAY
HORMONES & OTHERS
Prolactin IRMA Tube II

-
RADIO IMMUNOASSAY
HORMONES & OTHERS
FSH

-
RADIO IMMUNOASSAY
TUMOR MARKERS
SCC

-
RADIO IMMUNOASSAY
TUMOR MARKERS
NSE

-
RADIO IMMUNOASSAY
TUMOR MARKERS
FERRITIN

-
RADIO IMMUNOASSAY
TUMOR MARKERS
CA 15-3

-
RADIO IMMUNOASSAY
TUMOR MARKERS
CA 19-9(2step)
-
RADIO IMMUNOASSAY
TUMOR MARKERS
CA 19-9(1step)

-
RADIO IMMUNOASSAY
TUMOR MARKERS
CA125 IRMA Tube II

-
RADIO IMMUNOASSAY
TUMOR MARKERS
Ultra PSA

-
RADIO IMMUNOASSAY
TUMOR MARKERS
Free PSA

-
RADIO IMMUNOASSAY
TUMOR MARKERS
PSA

-
RADIO IMMUNOASSAY
TUMOR MARKERS
CEA

-
RADIO IMMUNOASSAY
TUMOR MARKERS
AFP

-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
Anti-Tg
-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
Tg-s

-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
RIAKEY TSH IRMA Tube II

-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
T3-Uptake
-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
Free T4

-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
Free T3

-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
T4

-
RADIO IMMUNOASSAY
THYROIDS & AUTOIMMUNE
T3

-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HCV

-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HAV lgM

-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HAV lgG
-
RADIO IMMUNOASSAY
HEAPATITIS
HBeAg/Ab

-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HBc (RIA)

-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HBc lgM

-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HBc lgG
-
RADIO IMMUNOASSAY
HEAPATITIS
Anti-HBs

-
RADIO IMMUNOASSAY
HEAPATITIS
HBsAg
-
RADIO IMMUNOASSAY
HEAPATITIS
RIAKEY AFP IRMA Tube II

-
POINT-OF-CARE
DIAKEY COVID-19 Ag Rapid Test
DIAKEY COVID-19 Ag Rapid Test

-
POINT-OF-CARE
DIAKEY COVID-19 IgM/IgG Rapid Test
DIAKEY COVID-19 IgM/IgG Rapid Test

-
POINT-OF-CARE
DIAKEY DOA6 RAPID
DIAKEY DOA6 RAPID

-
POINT-OF-CARE
DIAKEY Syphilis RAPID
DIAKEY Syphilis RAPID

-
POINT-OF-CARE
DIAKEY Anti-HCV RAPID
DIAKEY Anti-HCV RAPID

-
POINT-OF-CARE
DIAKEY Anti-HIV 1/2 RAPID
DIAKEY Anti-HIV 1/2 RAPID

-
POINT-OF-CARE
DIAKEY HBsAg RAPID
DIAKEY HBsAg RAPID

-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY NSE ELISA

-
ENZYME IMMUNOASSAY
HORMONES & OTHERS
DIAKEY FSH ELISA

-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY Ferritin ELISA

-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY Free T3 ELISA

-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY Free T4 ELISA

-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY TSH ELISA

-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY T4 ELISA

-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY PSA ELISA

-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY Anti-HAV IgG ELISA

-
ENZYME IMMUNOASSAY
THYROID & AUTOIMMUNE
DIAKEY T3 ELISA

-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY CA125 ELISA

-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY CEA ELISA

-
ENZYME IMMUNOASSAY
TUMOR MARKERS
DIAKEY AFP ELISA

-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY H.Pylori IgG ELISA

-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY Anti-HIV 1/2 ELISA

-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY Anti-HCV 3.0 ELISA

-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY H.Pylori IgG ELISA

-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY Anti-HBs ELISA

-
ENZYME IMMUNOASSAY
HEPATITIS
DIAKEY HBsAg ELISA

NOTICE


SHINJIN MEDICS
B301, 138, Ilsan-ro, Ilsandong-gu, Goyang-si, Gyeonggi-do, 10442, Korea